Industries
Medical
JBRplas manufactures medical-grade plastic injection molds and molded components — FDA compliant resins, ISO 10993 biocompatible materials, clean room molding available.
Medical Device Plastic Injection Molding
Plastic components used in medical devices and diagnostic equipment must meet stringent biocompatibility, dimensional, and traceability requirements that go beyond standard industrial molding. JBRplas has established processes specifically for medical-grade injection molding — from material selection and mold design through to clean room molding and lot-controlled shipping.
Medical Components We Produce
Diagnostic & Laboratory Equipment
- Diagnostic cartridge housings
- Sample collection device components
- Lateral flow assay housing and frames
- Pipette tips and tube caps
- Centrifuge tube bodies and caps
- Reagent bottle caps and inserts
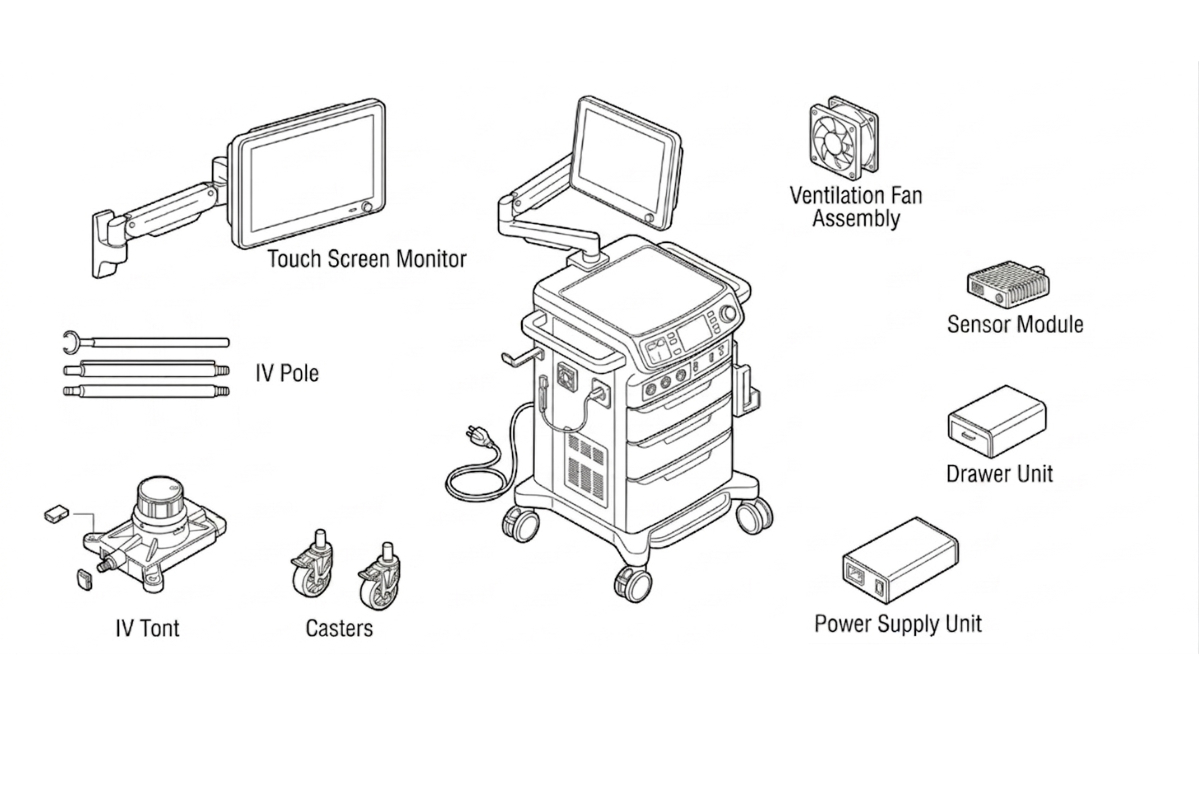
Patient-Facing Devices
- Respiratory mask frames and connectors
- Drug delivery device housings (inhaler, pen injector)
- IV administration set components
- Patient monitoring device enclosures
- Hearing aid housings
Surgical & Procedural
- Surgical instrument handles and grips
- Scope component housings
- Single-use procedure kit components
Dental & Orthopedic
- Dental tray and instrument handle components
- Orthopedic brace and support frame components
- Dental equipment housings
Biocompatible & FDA-Compliant Materials
| Material | Standard | Key Properties |
|---|---|---|
| PP (medical grade) | USP Class VI / FDA | Chemical resistance, steam sterilizable |
| PE (HDPE medical) | USP Class VI / FDA | Impact, inertness, chemical resistance |
| PC (medical grade) | USP Class VI / ISO 10993 | Clarity, autoclave resistance, impact |
| ABS (medical grade) | USP Class VI | Device housings, snap-fit enclosures |
| PEEK | ISO 10993, USP Class VI | High-temp sterilization, chemical resistance |
| TPE / TPU (medical) | ISO 10993, FDA | Soft seals, over-moulds, flexible connectors |
Clean Room Molding
JBRplas operates a Class 100,000 (ISO 8) clean room molding area for medical components:
- Dedicated clean room presses (60T – 250T)
- Gowning protocol for all personnel
- Positive pressure and HEPA filtration
- Particle count monitoring and logging
- Automated part handling inside the clean room
Traceability & Documentation
- Lot traveller — complete production record per lot
- Resin certificate — USP/ISO compliance per lot number
- Dimensional report — CMM measurement data per First Article
- Process records — injection parameters logged per production run
- Certificate of Conformance — per shipment, signed by quality manager
Other Industries We Serve
JBRplas manufactures precision plastic injection molds and molded parts for automotive OEM suppliers — interior trim, connector housings, HVAC components, and structural brackets.
Explore
Plastic injection molded enclosures, housings, and components for consumer electronics and home appliances. Flame-retardant resins, UL94 V-0, Class A finish. Shenzhen factory.
Explore